2020 Virtual AIChE Annual Meeting
(162v) Multi-Interpenetrating Polymer Networks (mIPNs) As 3D in Vitro Models to Evaluate the Effects of Hyaluronic Acid Degradation on Human Microglia Activation
Authors
Jolliffe, A. - Presenter, Trinity University
Contestabili, M., Trinity University
Cagle, T., Trinity University
Jimenez-Vergara, A., Trinity University
Munoz-Pinto, D., Trinity University
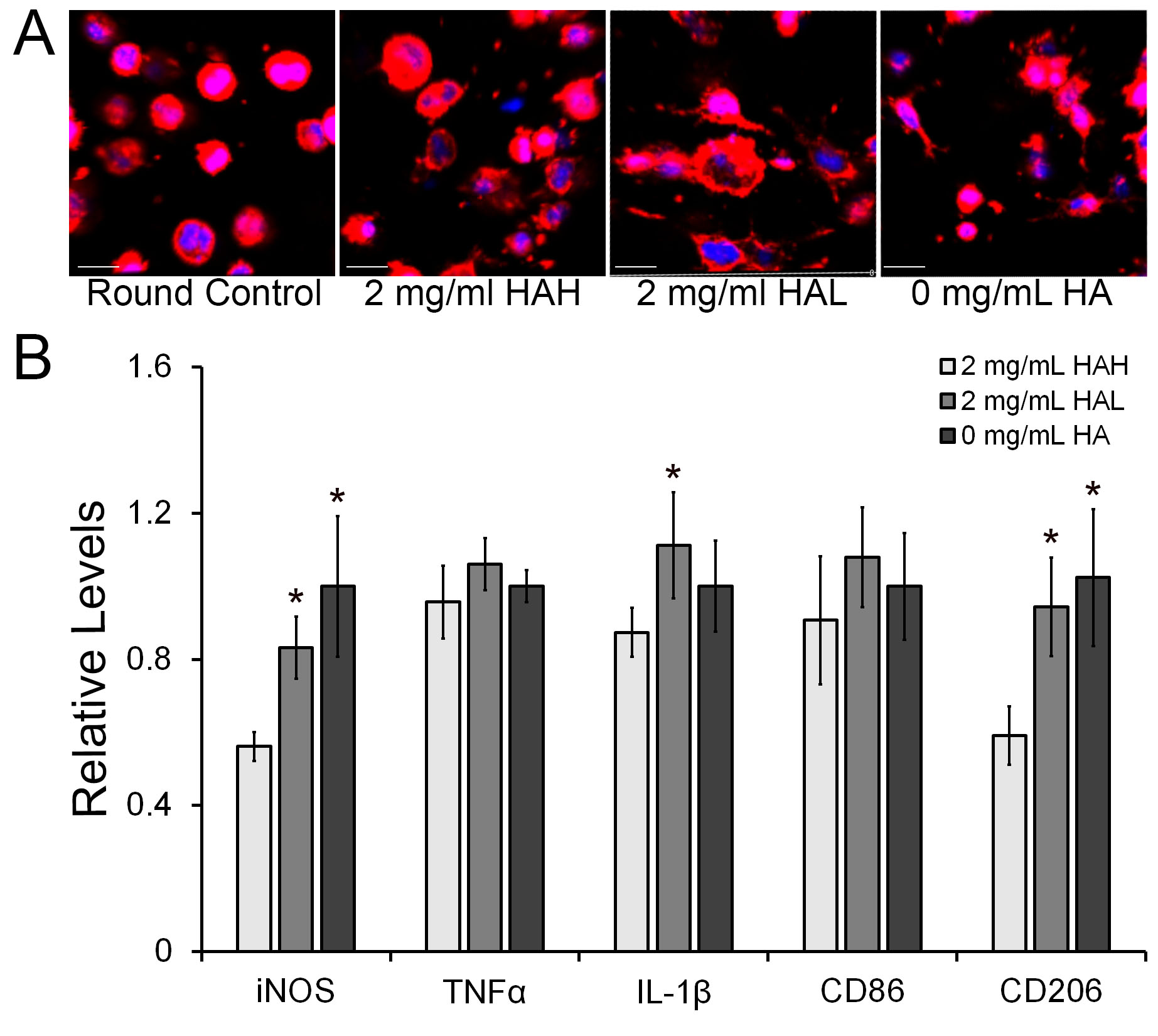
Background: Numerous recent studies have focused on the role of microglia in the onset and progression of neurodegenerative diseases including Alzheimerâs disease (AD) (Lee et. al. 2015). One of the areas of interest is the study of the physiological changes microglia elicit in response to neural damage. Macromolecules such as collagen type IV, laminin, and hyaluronic acid (HA) characterize the extracellular matrix (ECM) landscape of the central nervous system (CNS). These components contribute to the regulation of microglia activation and phenotype modulation. In response to tissue injury or trauma, the normal composition of the ECM can be altered to induce microglia activation. Methods. In this work, we modeled and evaluated the effects of HA molecular weight (MW) on human microglial cells (HMC3) under in vitro conditions using multi-interpenetrating polymer networks (mIPNs). We fabricated and characterized a family of mIPNs comprised of poly(ethylene glycol) diacrylate (PEGDA), collagen type I (Col I), and HA of high (HAH, ~1.5 MDa) or low (HAL, ~40 kDa) MW via sequential infiltration (Van Drunnen et al. 2019). The viscoelastic properties of mIPNs were evaluated using dynamic mechanical analysis (DMA). In these scaffolds, the effects of HA MW on microglia activation were studied independently of scaffold mechanical performance. After 3 days in culture, changes in the expression of microglia pro-inflammatory (M1) markers TNFâº, IL-1β, iNOS, and CD86, and the alternatively activated (M2) marker CD206 were measured at the protein level using immunofluorescence. Cell phenotypic expression at the protein level was quantified using confocal microscopy imaging and the ImageJ 2 (FIJI) program. Cell morphology was assessed using confocal microscopy. Results. The overall HMC3 viability in the mIPNs exceeded 85%. Microglia morphology appears to be modulated by HA MW. On day 3, the reduction in HA MW in the mIPN formulations promoted a significant increase in the expression of the M1 and M2 markers iNOS, IL-1β, and CD206 respectively. Conclusions. With the proposed engineered scaffolds, we demonstrated that the reduction in the MW of HA â which is normally associated with tissue degradation â is a significant contributing factor for human microglia activation.