2020 Virtual AIChE Annual Meeting
(488b) Including Anharmonicity in Adsorbate Partition Functions and How It Affects Theoretical Parameter Estimates for Desorption of Methanol from a Cu(111) Surface
Authors
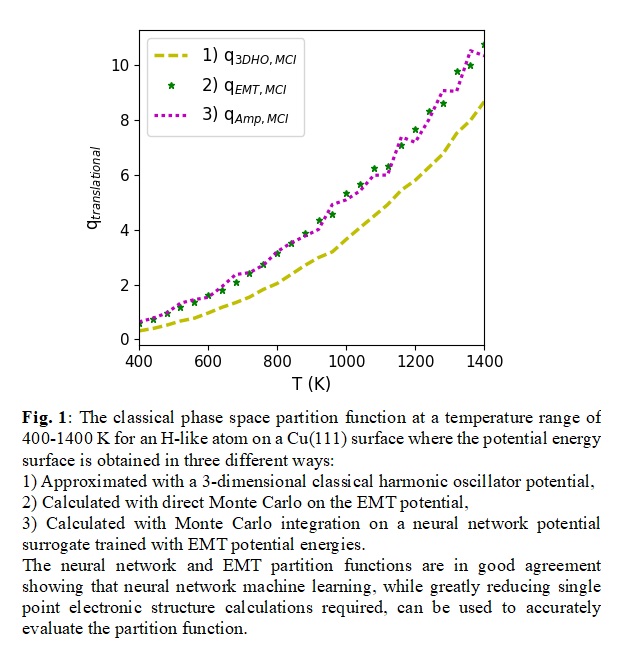
To compute the six-dimensional phase-space integrals, we construct surrogates for the potential energy surface from prior density functional theory calculations. We compare a range of surrogate forms, from sparse polynomial expansions to artificial neural networks, while the integration is performed either using quadrature or via Monte Carlo sampling. The accuracy and efficiency of the methods is assessed, along with an analysis of the importance of anharmonicity in microkinetics.
Obtaining an accurate partition function for methanol on Cu(111) is especially important for calculating the equilibrium constant of the desorption of physisorbed methanol, which is a key reaction in methanol synthesis. The effect of anharmonicity on the equilibrium constant is considered as well as the accuracy and efficiency of the surrogate and integration approaches for this case. This work is a part of a broader project within the Exascale Catalytic Chemistry (ECC) group, led by SNL, which uses methanol synthesis on copper as a test system.
This work was supported by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, Chemical Sciences, Geosciences and Biosciences Division, as part of the Computational Chemical Sciences Program. Sandia National Laboratories is a multimission laboratory managed and operated by National Technology and Engineering Solutions of Sandia, LLC., a wholly owned subsidiary of Honeywell International, Inc., for the U.S. Department of Energyâs National Nuclear Security Administration under contract DE-NA0003525.