2020 Virtual AIChE Annual Meeting
(513f) Sustainable Solid Acid Carbon Catalysts from Renewable Biomass for Fine and Specialty Chemical Synthesis
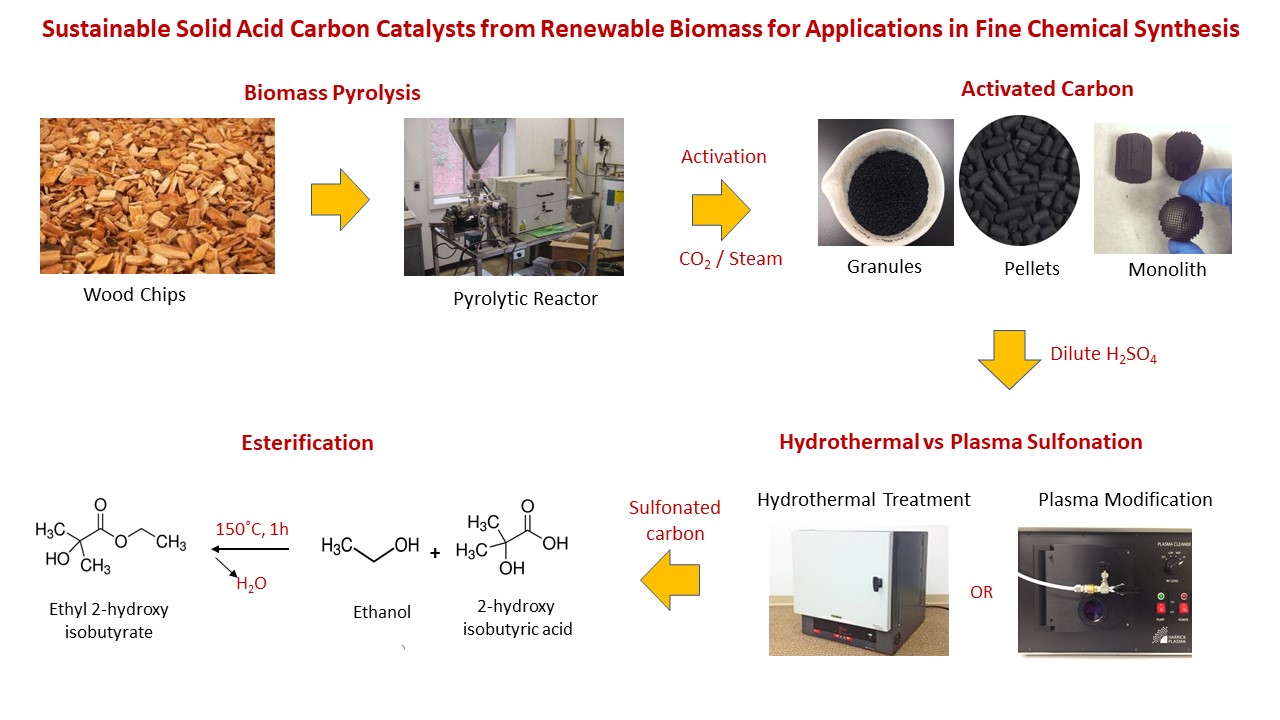
With the intent of incorporating sustainability into our catalyst preparation methods, activated carbon derived from woody biomass, in its granular and monolith forms was used to synthesize sulfonated carbon. Activated carbon monolith (ACM) confers the advantage of continuous processing with lower pressure drop and higher mass transfer rates over the granular activated carbon(GAC). We propose a new method where dilute H2SO4 can replace concentrated acid as the sulfonating agent. Innovations in green catalyst design have suggested that plasma technology could be a valuable tool for catalyst synthesis. This technology permits the synthesis of carbon catalysts at low temperatures and pressures, besides reducing the time required for catalyst preparation. Using plasma with different carrier gases, we demonstrate the functionalization of sulfonic acid on activated carbon in granular and monolith forms. The catalytic activity of the hydrothermal and plasma sulfonated carbon catalysts (GAC and crushed ACM) will be compared to a petroleum-derived, industrial benchmark catalyst, Amberlyst-15 in batch esterification reactions of 2-hydroxyisobutyric acid (2HIBA). This research could have potential implications in the catalytic upgrading of fermentation derived organic acids.