(85b) Sodium Tungstate-Promoted Perovskite Oxides As Effective Redox Catalysts for Redox Oxidative Cracking of Naphtha
AIChE Annual Meeting
2020
2020 Virtual AIChE Annual Meeting
Topical Conference: Next-Gen Manufacturing
Process Intensification and Modular Manufacturing: Chemical Commodity Processes
Monday, November 16, 2020 - 8:15am to 8:30am
We propose a redox oxidative cracking (ROC) approach as an alternative scheme to intensify the production of light olefins from naphtha. ROC avoids direct contact between gaseous oxygen and naphtha by cracking the vaporized naphtha feed, forming light olefins and hydrogen. Meanwhile, the hydrogen is selectively combusted by the lattice oxygen of a solid metal oxide redox catalyst to form water. The reduced redox catalyst is then re-oxidized with air in a separate regeneration step, completing the redox cycle. Here, we report Na2WO4-promoted perovskites, e.g. CaMnO3, as effective redox catalysts for ROC of naphtha. Using n-hexane and cyclohexane model compounds, over three-fold increase in olefin yield was achieved in the presence of the redox catalyst compared to thermal cracking. The redox reaction pathway, phase transition behavior of the perovskite oxides, and the role of Na2WO4 promoter were characterized in detail, providing important mechanistic insights for further optimization.
CaMnO3 was prepared from metal nitrates, citric acid, and ethylene glycol via a modified Pechini method. 20 wt.% Na2WO4 was added using wet impregnation. N-hexane and cyclohexane ROC tests used a fixed-bed quartz U-tube reactor and an automated gas delivery panel with downstream gas chromatography (GC) and mass spectrometry (MS). Samples were characterized via oxygen temperature-programmed desorption (O2-TPD), X-ray diffraction (XRD), in-situ XRD, X-ray photoelectron spectroscopy (XPS), low-energy ion scattering (LEIS), transition electron microscopy (TEM) with EDS mapping, FTIR spectroscopy, differential scanning calorimetry (DSC), and electrochemical impedance spectroscopy (EIS).
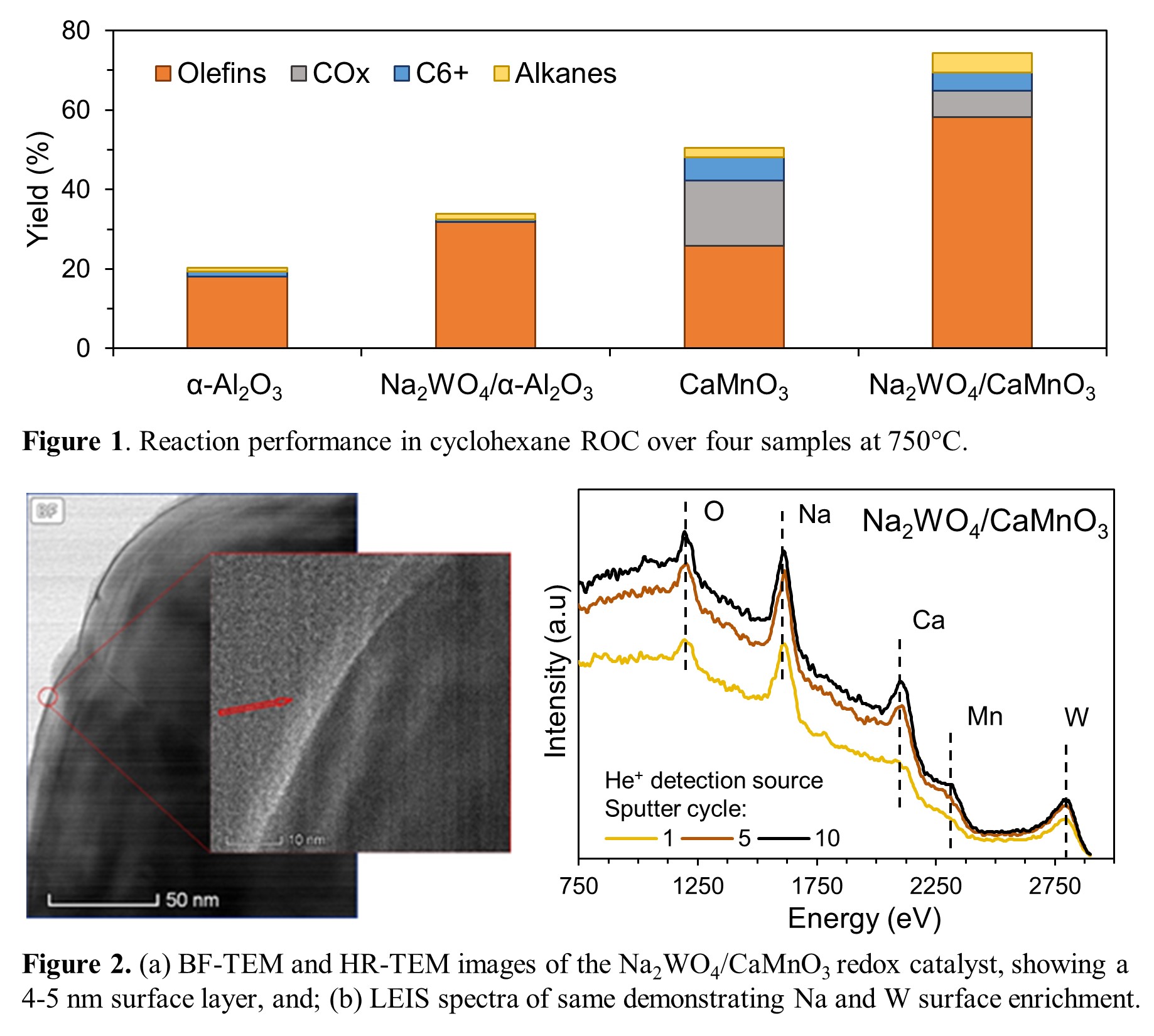
A 20 wt.% Na2WO4/CaMnO3 redox catalyst demonstrated 64% and 58% olefin yields from n-hexane and cyclohexane feeds, respectively, at T = 750°C and GHSV = 4500–5400 h-1, representing absolute single-pass olefin yield increases of 12% and 40% over thermal cracking of each compound at identical conditions (Figure 1). COx yield as low as 1.7% was achieved along with >80% H2 combustion over 25 redox cycles. Multiple characterizations including TEM-EDS and LEIS (Figure 2) show the existence of a Na2WO4 surface layer and a Na- and W-enriched shell around the CaMnO3 core. Moreover, in situ XRD and TGA-DSC indicate that the Na2WO4 surface layer forms a molten shell covering the CaMnO3 core under the working state of the redox catalyst. Na2WO4 played a key bifunctional role as (i) a catalytically active surface layer for hexane conversion and (ii) an O2-/e- conductor facilitating in-situ combustion of H2 by CaMnO3 lattice oxygen at the gas-molten shell interface.
Core-shell structured Na2WO4/CaMnO3 was an effective redox catalyst for enhanced olefin production. Aspen Plus simulation of naphtha ROC demonstrated 52% energy savings and 50% reduction in CO2 emissions on a per-ton ethylene basis compared to naphtha steam cracking.