(357o) Demonstrating the Impact of Shear and Surface Roughness on Thrombosis in Ventricular Assist Devices
AIChE Annual Meeting
2022
2022 Annual Meeting
Meet the Candidates Poster Sessions
Meet the Industry Candidates Poster Session: Pharmaceutical Discovery, Development and Manufacturing Forum
Tuesday, November 15, 2022 - 1:00pm to 3:00pm
Continuous-flow left ventricular assist devices (VADs) are implanted in end-stage heart failure patients to assist with blood circulation. However, VAD implantation often causes thrombosis i.e., blood clot formation, which results in complications such as device malfunction, invasive VAD replacement surgeries, stroke, and death. Several studies over the past three decades have elucidated components of VAD implantation that contribute toward thrombus formation, which have been famously defined as Virchow’s triad, that is, blood-surface contact, blood flow dynamics and innate biochemical factors impacting blood coagulability.
When blood contacts implanted surfaces, which in the case of VADs are often made of titanium alloys or polymers, plasma proteins adsorb to the surface, creating an adhesive layer for platelets i.e., thrombocytes to bind to with surface receptors. Therefore, implanted surfaces serve as a nidus for the formation of potentially dangerous thrombi, comprised mainly of platelets and a network of plasma proteins such as fibrinogen and von Willebrand factor (vWf).Thrombosis is driven by the aggregation of active platelets with plasma proteins and each other. Platelets may exist in either resting or activated states, the latter of which have higher protein binding affinities due to increased expression of surface receptors. With these receptors, platelets bind to key free-flowing proteins, fibrinogen and vWf, which exist in globular conformations when freely flowing in blood. Shear rates as low as 50 s-1 induce fibrinogen unfolding and expose binding sites for platelets. However, due to the weak nature of the fibrinogen-platelet receptor bonds, the number of platelets binding to fibrinogen markedly drops after 1000 s-1. On the other hand, vWf is a larger multimeric protein that elongates only at shear rates above 1000 s-1, with the largest multimers elongating at 5000 s-1. vWf forms stronger bonds with platelets than fibrinogen, therefore regions above 5000 s-1 support stable platelet aggregates. These well-established findings highlight the vital biophysical role played by physiological shear rates (<2000 s-1) and non-physiological shear rates (>2000 s-1) that are often applied by VAD pumps.
Some research has been done to elucidate the impact of varying surface topographies on platelet adhesion and/or surface protein adsorption; however, the conclusions are widely distributed and contradictory. Reported results range from a 150% decrease to a 400% increase in platelet adhesion on sub-micron scale roughened surfaces. This could be attributed to many parameters in the studies, ranging from roughness characterization to the scales or aspect ratios of topographical features. A majority of studies we found report roughness in terms of either arithmetic mean height (Ra) or contact angle. While the former indicates feature size in a simplified manner, the latter can be misleading on surfaces with nanoscale features. Furthermore, some studies were performed under static conditions whereas others involved hemodynamic flow conditions, thus not capturing flow-induced protein interactions with both the surface and platelets.
To that end, this work was designed to determine the impact of micro- and nanoscale titanium alloy surface topographies on platelet adhesion and platelet aggregate properties under various physiological and VAD-relevant shear rates using microfluidic devices. We used fluorescent microscopy to visualize surface adhesive platelets in real time, which were then quantified using an image processing algorithm. Additionally, part of the study was aimed toward systematically describing surface roughness using surface spatial frequencies to make the findings adaptable to more blood- biomaterial contact applications.
Methods
To investigate the phenomenon of shear-induced thrombosis on various surface topographies, we incorporated a set-up with a polydimethylsiloxane (PDMS) microfluidic device clamped on to a roughened titanium-alloy surface. Polished Ti-alloy surfaces were roughened to arithmetic mean height (Ra) values of 0.3, 0.8 and 2.1 μm using abrasive and sandblasting techniques. A mirror-finished surface with no additional polishing was used as a negative control. Three-dimensional surface profiles were obtained via laser scanning profilometry, and a custom MATLAB program was written to calculate the Ra values and generate mean Fourier transform energy profiles. We designed and fabricated microfluidic devices to precisely control the flow conditions and surface chemistry, while visualizing thrombus formation at the microscale. The microchannels had rectangular geometries with low Reynolds and high Peclet numbers so as to apply constant, well-defined wall shear rates over the length of the channel. Fluid shear rates of 1000, 2000 and 5500 s-1 were applied by varying the input flowrate, controlled by a syringe pump.
The perfusate was a blood analog comprised of platelet-rich plasma and hemoglobin-depleted red blood cells, thereby creating an optically clear suspension with similar composition and rheological properties as whole blood. The platelets were labeled with a fluorescent dye named mepacrine (quinacrine dihydrochloride), which combined with the suspension’s optical clarity, enabled the visualization of platelet adhesion and aggregation, and thus thrombus formation, via upright fluorescent microscopy over 15-minute durations. The entire system was maintained at a temperature of 37˚C to mimic physiological cellular mechanotransducive responses.
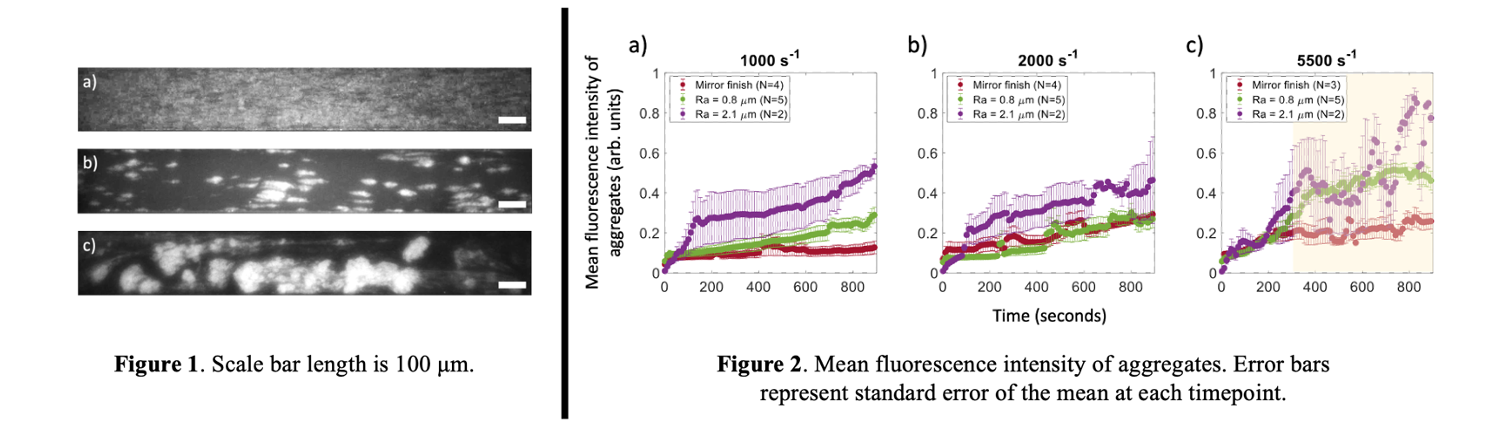
Images acquired via fluorescent microscopy were computed using a custom MATLAB program that included flat-field illumination correction, noise removal using pixel-wise low-pass temporal filtering, and binarization using Otsu dual-thresholding methods. From the binarized images, object (aggregate) properties were obtained, namely individual aggregate area, number of aggregates per frame and eccentricity of aggregates (not shown here). Additionally, mean object fluorescence intensities were plotted to compare aggregate densities across the parameter space (fig 2).
Results
At 1000 s-1, adhesion patterns demonstrated sustained growth of platelet monolayers (fig 1a) when Ra<2.1 μm. At 2000 s-1, stable, growing platelet aggregates were observed when Ra<2.1 um (fig 1b). At 5500 s-1, round platelet aggregates formed on all three surfaces, often connected by fibrous structures (fig 1c), with increasing intensity and density with time. Embolization was observed on all surfaces after an initial increase in aggregate intensity (in yellow, fig 2c). On the Ra 2.1 µm surface, platelet aggregates formed at all three shear rates, indicative of higher shear rates and gradients as a result of the roughness.
Future work will incorporate further analysis of platelet aggregate properties such as individual aggregate count, mean aggregate area and eccentricity. The former two properties determine the distinction between nucleation and growth of aggregates and the nature of aggregate growth; i.e., shape and direction of platelet deposition. Furthermore, these properties will be examined in conjunction with the spatial frequencies associated with the various surface topographies.
Implications
At both physiological and non-physiological shear rates, a surface with a 2.1 μm Ra causes thrombosis. Under non-physiological VAD-relevant shear, roughness leads to the embolization of thrombi, a phenomenon that has previously not been observed in studies investigating surface effects on thrombosis. These results demonstrate that rough surface topographies synergize VAD shear-induced thrombosis and thromboembolism. More broadly, these findings highlight the importance of polishing device surfaces to prevent dangerous and potentially fatal health complications.
Research Interests
My research interests are centered around using micro-scale mechanical tools and imaging capabilities to observe, understand and target mechanotransducive cellular processes in the human body. My graduate research has been focused on the mechanism known as thrombosis, i.e., blood clot formation, in the context of VADs, as described above. In the next phase of my graduate work, I will focus on leveraging microscopy and image processing tools in addition to microfluidics to visualize the entrapment and distribution of red blood cells during thrombus formation, and subsequently, how they impact the clinical dissolution of thrombi. Similarly, upon completing my graduate studies, my goal is to develop and improve upon implantable devices and diagnostic tools in the ever-evolving field of healthcare.