2022 Annual Meeting
(668b) Engineering Hydrogel Biomaterials with Tunable Mechanics and Proteolytic Stability Using Peptide Stereochemistry
Authors
Israt Jahan Duti - Presenter, University of Virginia
Jonathan Paul, University of Virginia
Emma Laudermilch, University of Virginia
Rachel Letteri, University of Virginia
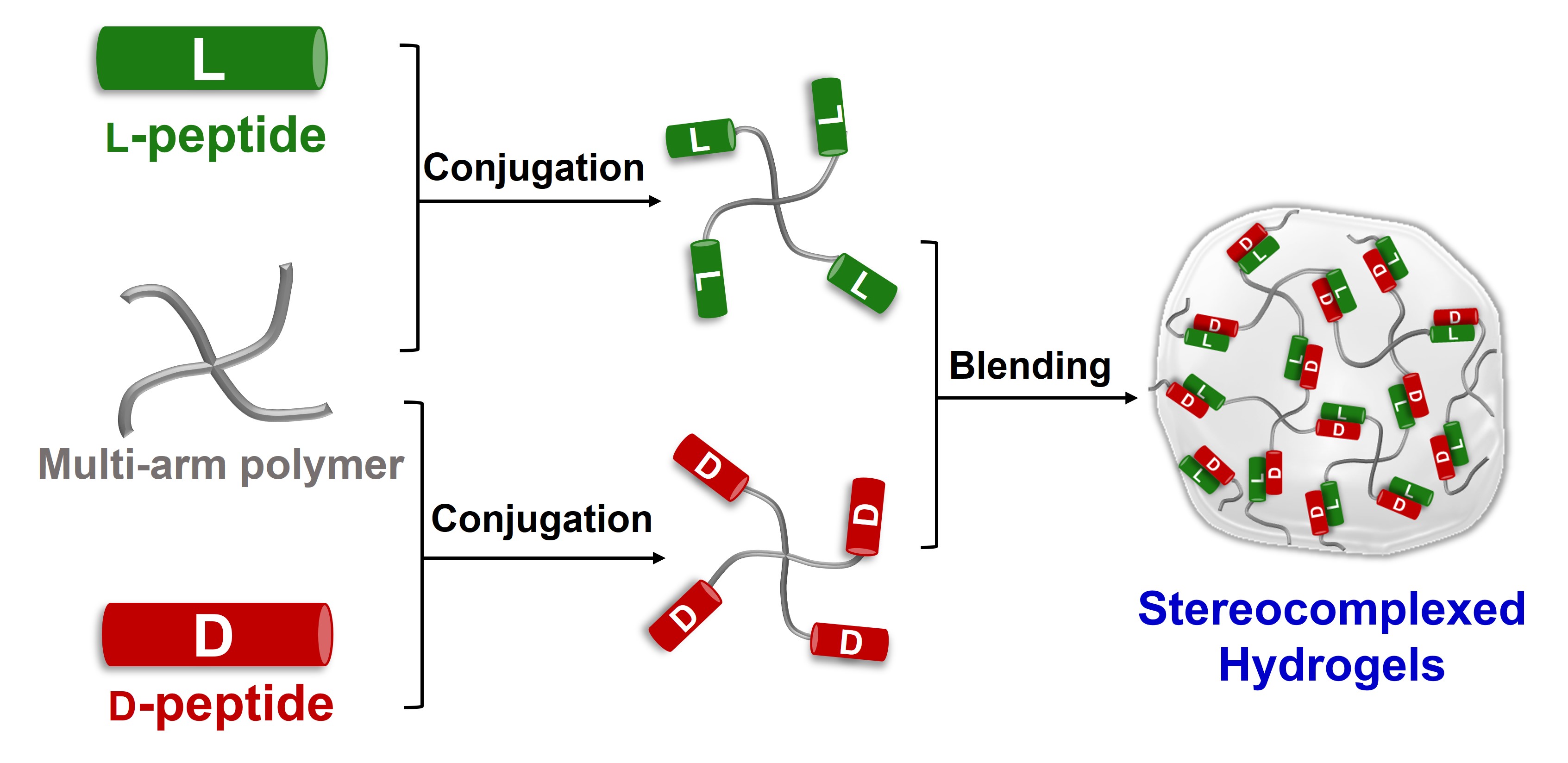
For tissue engineering and drug delivery, controlling the mechanics and lifetime of biomaterials is important to mimic native cellular environments and to modulate drug release rates. Leveraging stereochemistry-directed interactions between d- and l-peptides, we are developing hydrogel biomaterials with tunable mechanics and proteolytic stability from peptide-polymer conjugates. In these materials, assemblies of D- and L-peptides (stereocomplexes) serve as crosslinks, allowing us to mix solutions of complementary conjugates to generate hydrogels. We sought to learn how the molecular details of the peptides (length, charge, hydrophobicity, secondary structures) impact the properties of hydrogel biomaterials. Attaching fibril-forming β-sheet peptides (KYFIL) to 4-arm poly(ethylene glycol) produced conjugates that remain soluble up to 40 wt% when containing only D- or L-peptides. However, blending D- and L-conjugates in a 1:1 volumetric ratio at 10 wt% resulted in immediate hydrogel formation. Rheology showed the hydrogel storage modulus to be approximately 1 kPa, well suited for neural tissue engineering. We next analyzed a series of helical polyalanine-based conjugates with peptide sequences (AAXAA)n (A= alanine, X= cationic lysine or anionic glutamic acid, n=number of repeats from 3-5), varying the length and charge of the peptides. Even the conjugates with longest peptides having the complementary electrostatic interactions and opposite stereochemistry did not gel at 40 wt%. Hypothesizing the water solubility of the peptides may hinder their interactions in water, we introduced hydrophobic leucine residues to the polyalanines, and synthesized conjugates decorated with D- and L-(LAXLA)n. When X=lysine, blending D- and L- conjugates yielded hydrogels with 20 kPa storage modulus, suitable for muscle tissue engineering. We note that stereochemistry-driven crosslinks formed form both helical and β-sheet peptides, even in the presence of repulsive electrostatic interactions. Moreover, we found stereocomplexation to slow proteolytic degradation, allowing us to control biomaterial lifetime. In the future, we look forward to studying different peptide sequences as stereocomplex crosslinks to develop a new class of advanced biomaterials with tunable mechanics and proteolytic stability for cellular therapeutics and tissue engineering.