2023 AIChE Annual Meeting
(111h) Electrocatalytic Conversion of Phenol to Cyclohexane
Authors
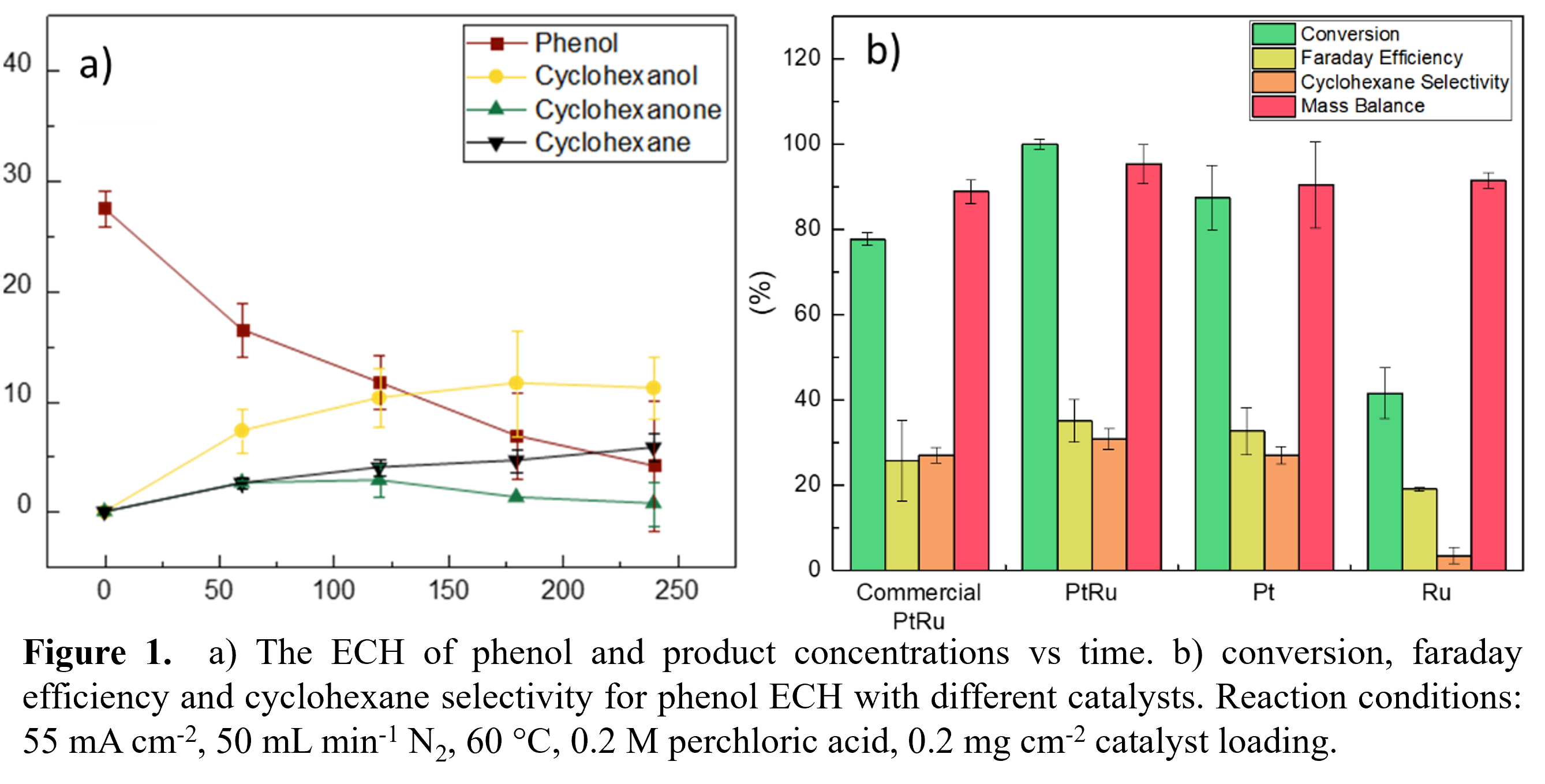
This work presents the upgrading of phenol as a bio-oil model compound. ECH of phenol was investigated in a custom electrochemical cell (H-Cell), wherein the anode and cathode are separated by a Nafion® 117 membrane. Preliminary ECH results using a commercial PtRu-C catalyst showed that after 4 hours of electrolysis at 55 mA cm-2, phenol conversion reaches 80%. The primary products of phenol ECH are cyclohexanol and cyclohexane with selectivity of 59% and 27%, respectively. Further ECH experiments were performed using lab synthesized PtRu, Pt and Ru catalysts to investigate the role of active metal on cyclohexane selectivity. These results (figure 1b) showed that the lab synthesized PtRu catalyst produced the highest cyclohexane selectivities (31%), highest conversion (100%) and efficiencies (35%). ECH of phenol was further investigated using operando Raman spectroscopy to understand the mechanism of cyclohexane formation. Preliminary results from these experiments showed that the cleavage of the C-OH bond depends on the potential.
(1) Zhang, X.; Lei, H.; Chen, S.; Wu, J. Catalytic Co-Pyrolysis of Lignocellulosic Biomass with Polymers: A Critical Review. Green Chemistry. 2016. https://doi.org/10.1039/c6gc00911e.
(2) Zhang, B.; Zhong, Z.; Min, M.; Ding, K.; Xie, Q.; Ruan, R. Catalytic Fast Co-Pyrolysis of Biomass and Food Waste to Produce Aromatics: Analytical Py-GC/MS Study. Bioresour. Technol. 2015, 189. https://doi.org/10.1016/j.biortech.2015.03.092.
(3) Page, J. R.; Manfredi, Z.; Bliznakov, S.; Valla, J. A. Recent Progress in Electrochemical Upgrading of Bio-Oil Model Compounds and Bio-Oils to Renewable Fuels and Platform Chemicals. Materials (Basel). 2023, 16 (1), 1â33.
(4) Chen, G.; Liang, L.; Li, N.; Lu, X.; Yan, B.; Cheng, Z. Upgrading of Bio-Oil Model Compounds and Bio-Crude into Biofuel by Electrocatalysis: A Review. ChemSusChem. 2021. https://doi.org/10.1002/cssc.202002063.