2023 AIChE Annual Meeting
(391g) A Novel Approach for Direct and Continuous Electrochemical Manufacturing of Lithium Hydroxide from Geothermal Brines
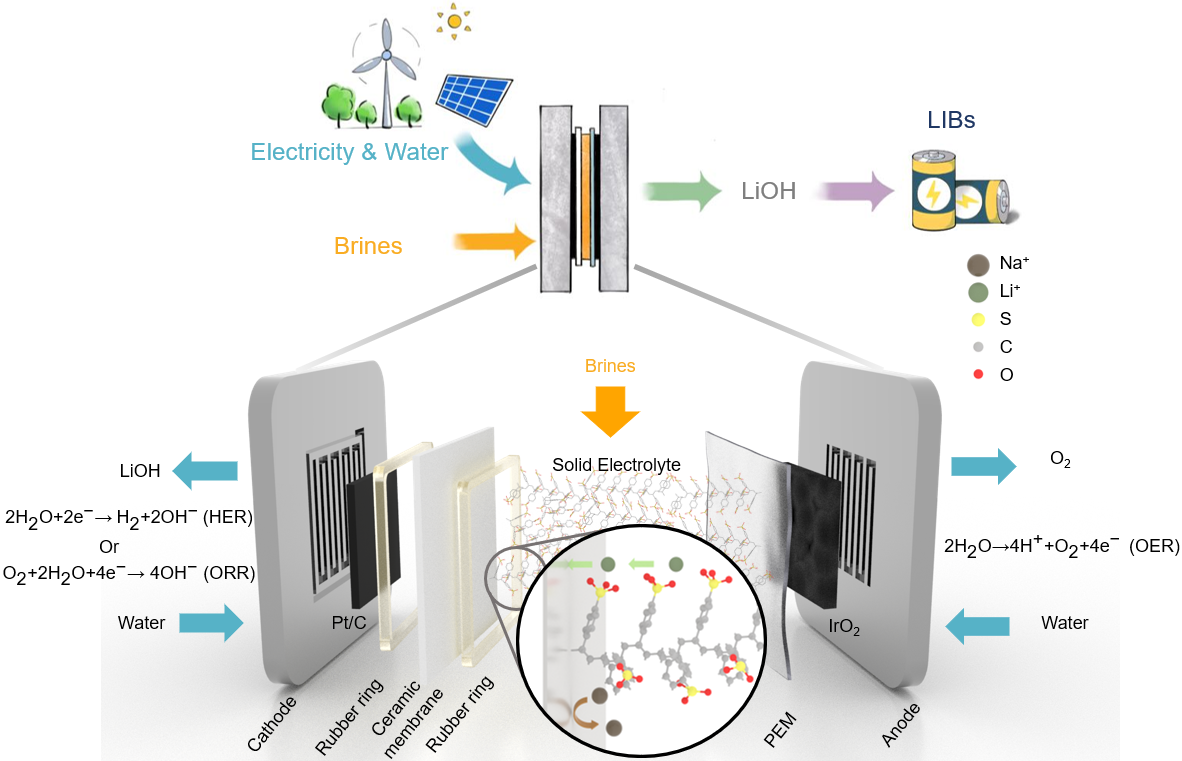
Herein, we present an effective and sustainable strategy for electrochemical lithium recovery from geothermal brines. Our approach utilizes a three-chamber solid electrolyte (SE) electrochemical reactor with a lithium conductive ceramic selective membrane (LISICON). As depicted in Fig 1, the simulated brine containing LiCl and NaCl flows into the middle layer filled with SE, while water flows in both the anode and cathode. The functionalized SE with sulfonate groups guarantees efficient Li+ and Na+ conduction, while only Li+ could transport through the lithium-ion conductive ceramic membrane, and combine with the OH- generated from HER. Starting from the simulated brine with initial Li: Na ratio of 1:100, we achieved a high Faradaic Efficiency (FE) of 97.51 ± 3.2%, based on Li+ concentration, and a Li+/Na+ selectivity ratio of over 2×103. Our device continuously recovers LiOH from brine with less contamination of Cl2 and HClO, and the average cell voltage corresponds to energy consumption of 9.87 kWh/kgLi, which represents a carbon footprint of 8.39 kgCO2e/kgLiOH. The presence of Na+ would lead to an increase of cell voltage and partially unreversible membrane fouling, but the ceramic membranes can be regenerated partially by operating with LiCl (1 M) solution in the middle layer. With ion chromatography (IC) analysis of the catholyte and Time-of-Flight Secondary Ion Mass Spectrometry (TOF-SIMS) analysis on the ceramic membrane, we confirmed that lithium is successfully recovered only from the simulated brine.