(521en) Repurposing Existing Steam Reformers for COX-Free Hydrogen Generation through Catalytic Ammonia Decomposition
AIChE Annual Meeting
2023
2023 AIChE Annual Meeting
Catalysis and Reaction Engineering Division
Poster Session: Catalysis and Reaction Engineering (CRE) Division
Wednesday, November 8, 2023 - 3:30pm to 5:00pm
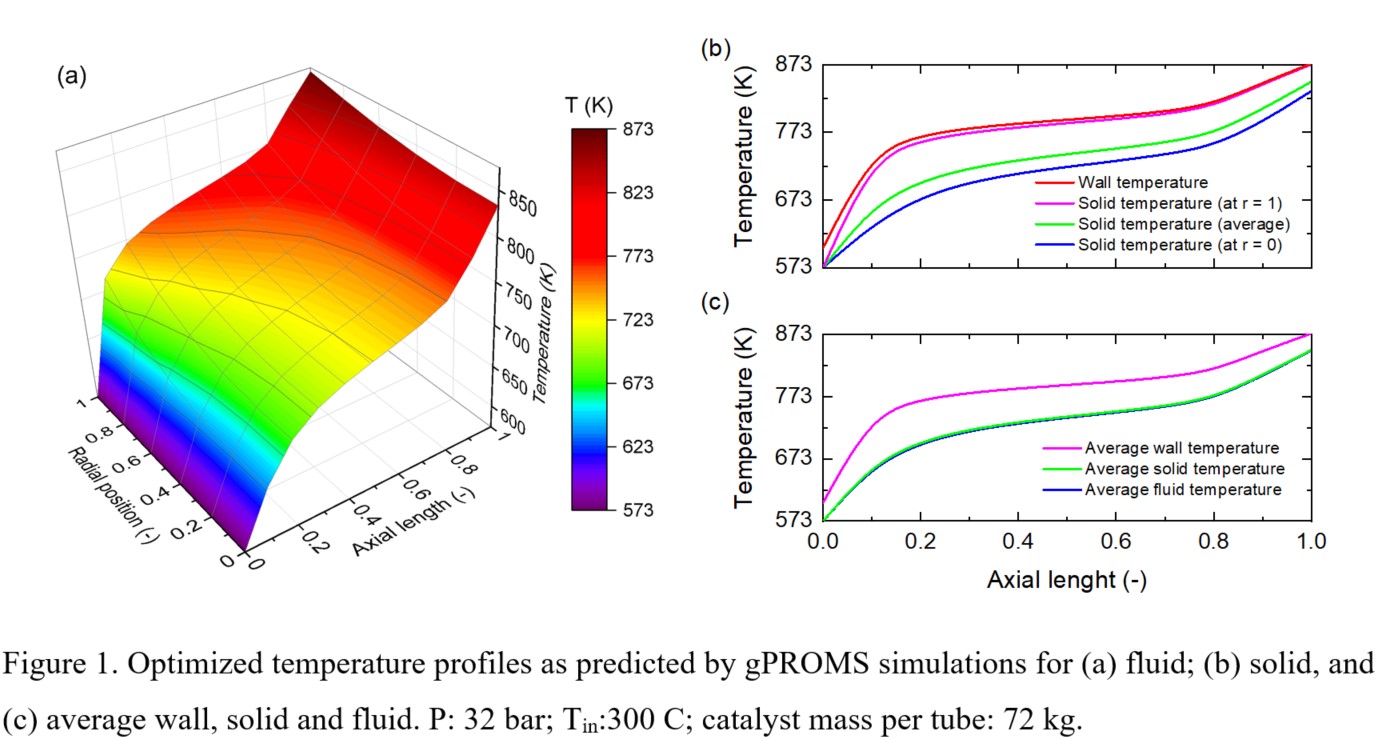
Towards that effect, we present a combined process simulations and computational fluid dynamics (CFD) approach to repurpose an industrial steam reformer as an ammonia cracker. The proposed reformer is modeled in a shell-and-tube configuration, with a Ba-Co-Ce catalyst loaded in multiple tubes and heated from the shell. Firstly, laboratory-scale ammonia decomposition runs were performed in a packed-bed reactor from which intrinsic kinetics were obtained as a modified power-law rate expression. Coupling them with a 2D cylindrical catalyst pellet incorporating heat-mass transfer effects in a cracker tube, process optimization is performed using gPROMS®. The optimization studies suggest using a profiled wall temperature to effectively use the entire tube length; avoiding cold spots (Figure 1). In the following stages, LES-based CFD simulations are performed using Barracuda Virtual Reactor® to further propose effective temperature control strategies for this endothermic reaction.