2023 AIChE Annual Meeting
(524d) Separation of Dihydroxyacetone from Glycerol Catalytic Oxidation Products in a Simulated Moving Bed Cascade: From Concept to Proof?of?Concept
Authors
Introduction
The biodiesel industry is growing as a response to the actual climate crisis and fossil fuel depletion, enhanced also by international policies1, leading to a surplus of glycerol (GLY), the main reaction by-product, which represents about 10 wt% of the production. GLY valorization by other routes rather than by incineration is important to increase biodiesel industry competitiveness and sustainability1.
Catalytic oxidation is one of the several GLY well-known valorization routes, yielding several high-added-value products, among them dihydroxyacetone (DHA), with major applications in the cosmetics industry, namely in sunless skin tanning lotions1. Several works concerning DHA production with high yields by the catalytic oxidation of GLY in the liquid phase using heterogeneous catalysts and O2 as an oxidant may be found in the open literature1; however, as far as we know there are no works concerning DHA purification from GLY catalytic oxidation. The cosmetics industry requires DHA with a minimum purity of 97%, therefore, DHA must be separated from the unreacted GLY and reaction byproducts, mostly organic acids: oxalic acid (OXA), tartronic acid (TTA), glyceric acid (GCA), and glycolic acid (GCO).
Adsorption isotherms of GLY and its oxidation products were determined at 293 K on a commercial polystyrene-divinylbenzene ion-exchange resin functionalized with sulfonic groups, the Dowex® 50WX-2 in acid form. All compounds presented a linear adsorption isotherm (except for oxalic acid, the least retained compound), with DHA and GLY being the most retained species2. Adsorption isotherms of GLY and DHA in the Dowex 50WX-2 in calcium form were also determined at 293 K, showing linear adsorption isotherm and DHA being the most retained compound2.
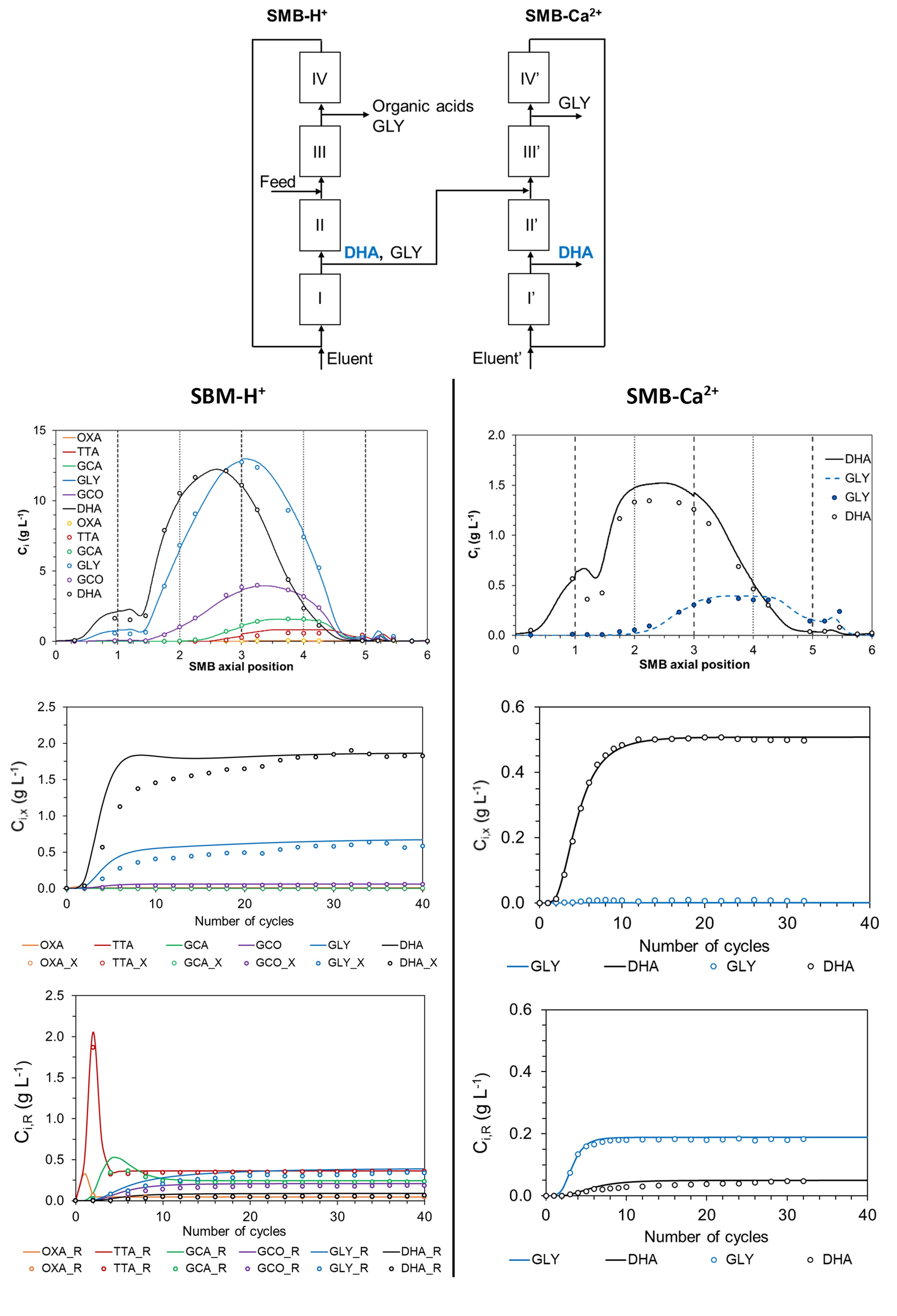
Due to the low selectivity for the DHA separation from GLY in the resin in acid form, (α = 1.04), compared with the selectivity in calcium form (α = 1.28), a DHA purification process by continuous chromatography is herein proposed on a cascade of two Simulated Moving Bed (SMB) units. DHA will be separated from the organic acids on an SMB packed with the resin in acid form (SMB-H+), being collected in the extract stream together with part of the GLY. This stream will be fed to a second SMB packed with the resin in calcium form (SMB-Ca2+) to separate DHA from GLY.
The process was implemented on the gPROMS model builder software V7.0.7 (PSE, UK) and it was experimentally validated on the FlexSMB-LSRE®, a lab-scale SMB unit with six fixed-bed columns packed with the Dowex® 50WX-2 resin.
The solution to be separated by the SMB cascade is the one obtained from the aerobic oxidation of a 1 M GLY aqueous solution with a commercial catalyst (Bismuth-doped Platinum nanoparticles supported in activated carbon) after 1.5 h in a semi-batch reactor, the most promising result among the platinum-based commercial catalysts tested2, with the following composition: 1.2 gOXA L-1, 12.9 gTTA L-1, 8.2 gGCA L-1, 6.9 gGCO L-1, 22.0 gGLY L-1, and 29.8 gDHA L-1.
Methods
Six stainless steel fixed-bed columns (100 x 20 mm) were packed with the Dowex® 50WX-2 resin and characterized (bulk porosity and Peclét number) by pulse injections of a tracer (Blue Dextran 2000) into the columns previously equilibrated in the eluent.
A mathematical model was developed to describe the fixed-bed column, considering the plug flow model with axial dispersion to describe the fluid flow, describing the mass transfer between the liquid and the solid by the linear driving force model, isothermal operation, homogeneous spherical particles with uniform size, constant fluid velocity, bed porosity, and bed length, and no bed radial gradients2. The model was implemented in the gPROMS model builder software and validated using single and multi-component breakthrough experiments. The model was extended to the SMB multicolumn process (described elsewhere3), and accounting with the FlexSMB-LSRE® tubes and equipmentâs dead volumes, column filters (fritz), manifolds, and time switch delays (switch asymmetries)4.
The operating conditions of the SMBs were defined by determining the separation and regeneration regions, where is the ratio between the liquid and the solid flow rate in each SMB section. The regeneration regions were obtained by applying a safety factor of 15% to the given by the equilibrium theory3. The separation regions were obtained based on the FlexSMB-LSRE® model, considering a minimum extract purity and DHA recovery in the extract stream (defined elsewhere3).
SMB experiments were performed in the FlexSMB-LSRE® unit with the 1-2-2-1 configuration, and the switching time t*, which defines the time to switch the inlet and outlet ports, was determined by applying a safety factor to the minimum t* for each SMB3.
The six compounds were fed to the SMB-H+ and 5mM H2SO4 aqueous solution was used as eluent. At the end of every SMB cycle (6t*), the inlet flowrates (feed and eluent) were measured as they were in graduated cylinders, while the raffinate and the extract outlet streams were collected separately, weighted, and analyzed by High Pressure Liquid Chromatography (method defined elsewhere2). When the speciesâ concentration in both raffinate and extract streams did not change more than 5% during five successive cycles, cyclic steady state (CSS) conditions were reached. At this point, internal concentration profiles were obtained by collecting samples at 25%, 50%, and 75% of the t*. The extract stream of the SMB-H+ was fed to the SMB-Ca2+ to separate DHA from GLY, using water as the eluent, and following the same procedure.
A fast startup method5 was implemented on the SMB-H+, which allowed to reduce ten cycles to achieve CSS conditions, whereas its application on the SMB-Ca2+ experimental trial was irrelevant for the given operation point.
Results and Discussion
The columns packed with the resin in acid form showed an average ε of 0.365, whereas the ones packed with the resin in calcium form showed an average ε of 0.384. The columns showed an average Pe number of 600 for both resins.
A minimum DHA recovery in the extract stream of 85% and an extract purity on a GLY-free basis of 97% were considered for the separation region of the SMB-H+. The operation point γI* = 2.88, γII*= 2.44, γIII* = 2.47, γIV* = 1.41, and a t* of 1.5 min were defined. The unit reached CSS conditions after 30 cycles, with 87.0% of DHA recovery in the extract stream and an extract purity in GLY free basis of 96.8%. The speciesâ average concentration in the extract stream was 1.82 g L-1 of DHA and 0.58 g L-1 of GLY, and the main contaminant was GCO (0.06 g L-1, 2.3%), while the concentration of the remaining organic acids was below 0.2%.
A minimum DHA recovery in the extract stream of 90% and an extract purity of 97% were considered for the separation region of the SMB-Ca2+. The operation point γI* = 3.31, γII* = 2.59, γIII* = 2.81, γIV* = 2.07, and a t* of 2 min were defined. The unit reached CSS conditions were achieved after 30 cycles with a DHA recovery in the extract stream of 89.0% and an extract purity of 98.6%, and the species average concentration in the extract stream was 0.50 g L-1 of DHA and 0.01 g L-1 of GLY.
The model fitted well the internal concentration profiles and the speciesâ concentration in the outlet streams of both SMB experiments. The two-SMB cascade produced DHA with a concentration of 0.50 g L-1 at a flow rate of 4.5 ml min-1, with 98.6% of purity. The cascade showed a global productivity of 21.7 kgDHA (m3adsorbent·day)-1 and an eluent consumption of 5.7 m3adsorbent kgDHA-1.
Conclusions
The purification of DHA from the reactional mixture obtained during GLY aerobic oxidation with Pt-based catalysts was experimentally demonstrated on a lab-scale SMB unit, the FlexSMB-LSRE®. The FlexSMB model was validated together with the kinetic and equilibrium data previously obtained on the fixed-bed column.
The two-SMB cascade produced DHA with 98.6% of purity, showing a global productivity of 21.7 kgDHA (m3adsorbent·day)-1 and an eluent consumption of 5.7 m3adsorbent kgDHA-1.
References
- Katryniok, B.; Kimura, H.; SkrzyÅska, E.; Girardon, J.-S.; Fongarland, P.; Capron, M.; Ducoulombier, R.; Mimura, N.; Paul, S.; Dumeignil, F., Selective catalytic oxidation of glycerol: perspectives for high value chemicals. Green Chemistry 2011, 13 (8), 1960-1979.
- M. Walgode, P.; D. Coelho, L. C.; V. Faria, R. P.; E. Rodrigues, A. r., Dihydroxyacetone Production: From Glycerol Catalytic Oxidation with Commercial Catalysts to Chromatographic Separation. Industrial & Engineering Chemistry Research 2021.
- Rodrigues, A., Simulated moving bed technology: principles, design and process applications. 1st Edition ed.; Butterworth-Heinemann: Amsterdam, 2015; p 304.
- Gomes, P. S.; Zabkova, M.; Zabka, M.; Minceva, M.; Rodrigues, A. E., Separation of chiral mixtures in real SMB units: The FlexSMBâLSRE®. AIChE journal 2010, 56 (1), 125-142.
- Xie, Y.; Mun, S.-Y.; Wang, N.-H. L., Startup and shutdown strategies of simulated moving bed for insulin purification. Industrial & engineering chemistry research 2003, 42 (7), 1414-1425.