2023 AIChE Annual Meeting
(559b) Mechanistic Insights into the Catalytic Transfer Hydrogenation of Muconic Acid in Solvents
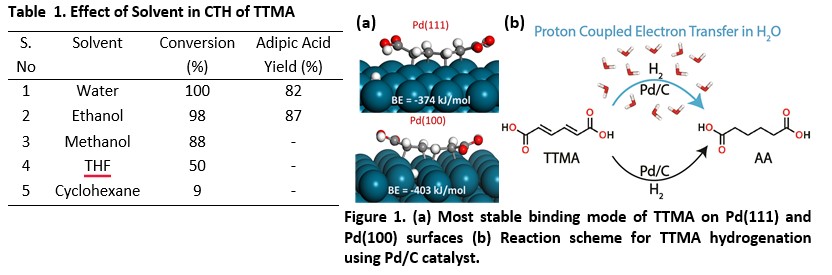
The transition metal-catalyzed hydrogenation of olefin functionality primarily occurs via the Langmuir Hinshelwood mechanism. The presence of solvents often tends to alter the conventional mechanism of hydrogenation, promoting alternate routes which are energetically more favorable.1,2 In the present study, catalytic transfer hydrogenation (CTH) of trans,trans-muconic acid (TTMA) to adipic acid (AA) using Pd/C catalyst and formic acid as a hydrogen source at 70°C and autogenous pressure is explored in different solvents. Interestingly, 100% conversion with 82% selectivity to AA is observed in water in 2 hrs (Table 1). Very high conversion is observed in polar protic solvents such as ethanol and methanol as well (Table 1). However, in THF, the conversion is observed to reduce to half, and in cyclohexane, the conversion drops to 9% (Table 1). Further, water THF solvent mixtures at different ratios were used to probe the mechanism of the hydrogenation reaction. We hypothesize that the hydrogen bonded network of water molecules assist the hydrogen transfer to the C=C bond via a proton coupled electron transfer mechanism (PCET). A detailed mechanistic study is carried out by modeling the system in different solvents and reaction environments using DFT simulations. Figure 1. shows the most stable binding mode of TTMA on Pd(111) and Pd(100) surfaces. The adsorption energy, activation barriers, and reaction energies of elementary steps is observed to change significantly in the presence of explicit solvents applied in the simulations. We observe a favorable energetics for the proton-coupled electron transfer mechanism for the hydrogenation of muconic acid in protic solvents as compared to that of the conventional hydrogenation route.