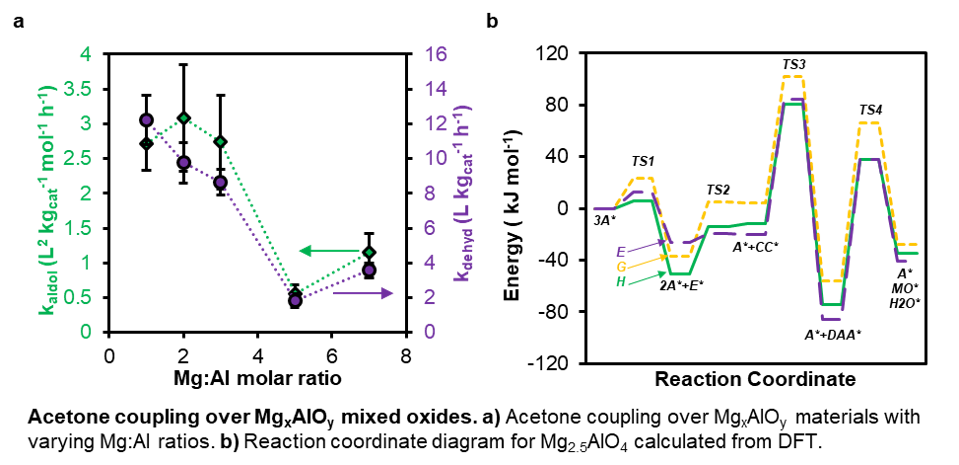2023 AIChE Annual Meeting
(616g) Understanding the Influence of Metal Composition on Aldol Condensation Reactions over MgxAlOy-Type Materials Via Combined Experimental and Computational Investigations
Authors
In this work we synthesized Mg-Al mixed oxides with varying Mg:Al molar ratios via the thermal decomposition of alkali-free layered double hydroxide precursors and examined them in the liquid-phase aldolization of acetone as a model reaction. Thermal and structural characterizations demonstrate the requirements for LDH decomposition without causing structural collapse. Reactivity trends and identification of active sites are examined via data-fitted reaction kinetics models and discussed in connection with ex-situ and in-situ site titration experiments as well as infrared spectroscopy and reversible poisoning experiments. Additional details are gathered via density functional theory (DFT) simulations of acetone aldolization over model MgxAlOy surfaces. These simulations suggest that the doping of Al creates metal vacancies which act as strong base sites. Consistent with experiment, these strongly basic sites are shown to be crucial to aldolization and help to explain the high sensitivity of aldolization rates to Mg:Al molar ratios in these mixed oxides. These studies guide future work toward developing materials comprising more exotic metal combinations in order to improve the selectivity and reactivity of heterogeneous aldolization reactions.