2023 AIChE Annual Meeting
(94g) One-Pot Synthesis of a Novel Chitosan-Based Hydrogel By C-Mannich Reaction and Screening of Its Cu (II) Uptake
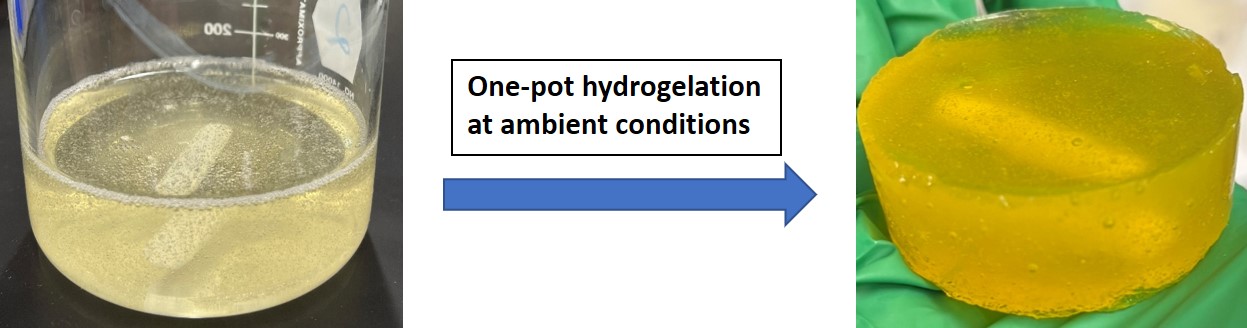
The persisting industrialization continues to worsen the dilemma of metal-induced water pollution. Water treatment by adsorption is known to be one of the most efficient methods, but the harsh manufacturing conditions of activated charcoal and the non-sustainability of hydrocarbon-based commercialized adsorbents imposed extra costs to this treatment strategy. More recent efforts are driven towards the utilization of polysaccharide-based hydrogel adsorbents as sustainable alternatives. This work presents the fabrication, characterization, and screening of Cu2+ uptake of a novel chitosan-based hydrogel achieved in a single step by C-Mannich reaction. The Mannich base crosslinked-chitosan hydrogel (CS-MB) was successfully fabricated in one-pot with chitosan (CS), glutaraldehyde (Glu), and 4-hydroxycoumarin (4-HC) in 3% v/v acetic acid at 22 0C.[1] The novel CS-MB exhibits a temperature and pH-dependent swelling behavior unique from other hydrogel systems with an optimal swelling at pH = 4 and 22 0C. Using this optimal swelling condition, Cu2+ uptake was conducted in batch mode and achieved a maximum adsorption capacity of 12.0 mg/g with 1.2 g/L adsorbent dosage after 36 h with agitation. The uniform surface adsorption of Cu2+ was verified by EDS and the adsorption site was identified by FT-IR. The one-pot fabrication method of the novel CS-MB at ambient conditions using a sustainable polysaccharide and its activity in Cu2+ uptake provides a promising cheap and sustainable alternative to the commercialized adsorbents for the removal of Cu2+ in water by adsorption.
References
[1] Romal, J.R.A*.; Ong, S.K. Single-Step Fabrication of a Dual-Sensitive Chitosan Hydrogel by C-Mannich Reaction: Synthesis, Physicochemical Properties, and Screening of its Cu2+ Uptake. Processes 2023, 11, 354. https://doi.org/10.3390/pr11020354