(275c) Intensifying the Recovery of Carboxylic Acids from Fermentation Broths Via Reactive Extraction inside the Membrane-Assisted Spinning Disc Reactor
AIChE Annual Meeting
2022
2022 Annual Meeting
Process Development Division
Process Intensification - Novel Reactors
Tuesday, November 15, 2022 - 8:50am to 9:15am
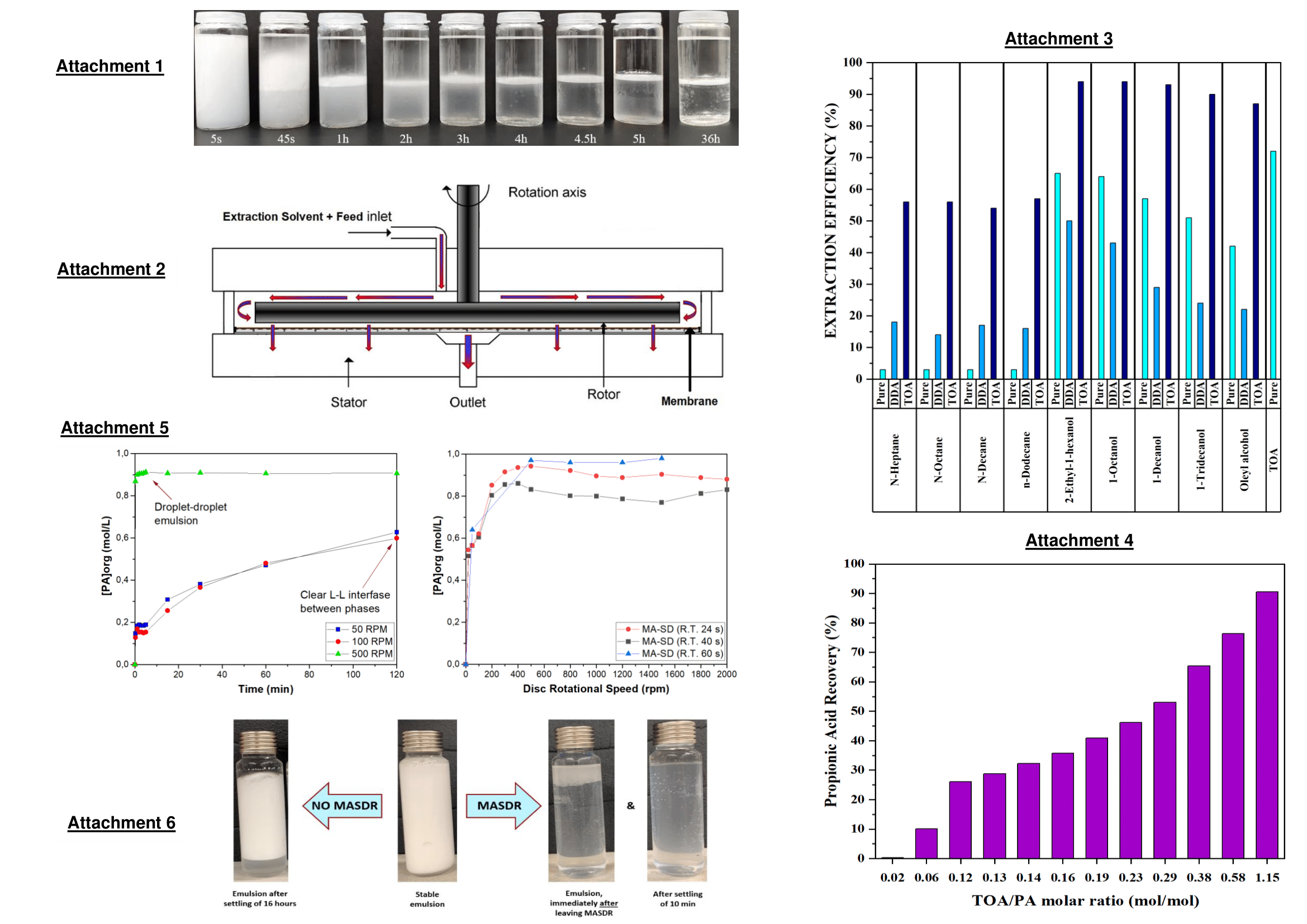
Here, the solvent and the feed are co-fed into the MASDR, where high shear is ensured to reach equilibrium conditions very fast. However, above a certain shear rate very stable emulsions would form, which would take very long times to phase-separate. By placing a membrane at the bottom side of the spinning disc, phase separation may be achieved by breaking the stability of emulsions due to forced transport through porous media, ultimately reaching demulsification. Extraction and phase separation may be thus achieved in the same equipment, making it promising for biotechnological applications.
Preliminary equilibrium studies for the extraction of propionic acid using pure solvents (i.e., solvent extraction), or combinations of a solvent with a tertiary amine (i.e. dimethyldodecylamine - DDA - or tri-n-octylamine - TOA -), were carried out in order to find the best extractive system for propionic acid recovery from synthetic fermentation broths (< 4.5 wt. %). The results of the equilibrium studies are presented in Attachment 3. The extractive system TOA in 1-Decanol was selected as the desired solvent for reactive extraction, and after having studied the effect of TOA/PA molar ratio on extraction efficiency, it was inferred that PA extractions above 75% can be achieved if only TOA/PA mol ratios are higher than 0.58 (see Attachment 4).
Batch and MASDR experiments were carried out at 35 ËšC for the recovery of propionic acid (0.6 mol/L), using a mixture of 30 vol.% TOA in 1-decanol. The aqueous-to-organic ratio (volume/volume) was two. The results are shown in Attachment 5.
In Attachment 5, it was concluded that the high shear induced by MASDR helped to quickly achieve equilibrium from 200 RPM onwards (i.e. droplet-droplet emulsion region), which reflects its superior performance in terms of mixing compared to batch systems. However, going above 500 RPM did not improve performance any further, even at residence times as low as 24 seconds. In fact, it should be prevented as higher shear rates generate much more stable emulsions which are extremely hard to separate. If these stable emulsions, once formed, were collected and re-entered the MASDR one more time at e.g. residence time of 1 min, partial demulsification can be quickly achieved after 10 min, as shown in Attachment 6. Phase separation is clearly visible, which does not occur in long-time ordinary settling, though full separation was still not reached for that timespan. By limiting MASDR operation below 500 RPM, the extent of demulsification may be much higher, optimizing the process. Further study on demulsification properties under those operating conditions is currently in progress, and a more detailed explanation will be given at the presentation.