Follow these eight steps to work effectively with your catalyst supplier.
Specifying the right catalyst bed can mean the difference between years of profitable and trouble-free operation and years of continuous problems. Yet questions surround every decision, from how to define the process requirements to estimating catalyst life.
The catalyst business is cloaked in mystery. End-users want the perfect catalyst — one that completely converts reactants with no byproducts and lasts forever, all at a low price; they treat the required conversion, acceptable selectivity, and manageable catalyst life like trade secrets that would reveal operational weaknesses. Catalyst suppliers are reluctant to disclose details of the catalyst’s properties, afraid that the competition will create cheaper copies. Engineering companies do their best to arbitrate the discussions and probe for the hidden relationship between aspects of performance and catalyst cost. Miscommunication is common at all stages of the catalyst specification process, thanks to this tradition of secrecy.
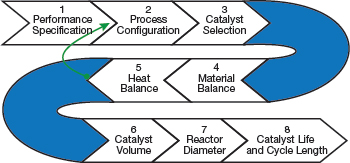
▲Figure 1. The eight steps to specifying a catalyst bed are determining performance specifications and the process configuration, selecting the catalyst, performing the material and heat balances, determining the catalyst volume and reactor diameter, and estimating the catalyst life and cycle length.
This article presents a step-by-step approach to specifying a hydrogenation catalyst (Figure 1).
Step 1. Performance specification
The first step of the procedure is communication between the catalyst vendor and the end-user or the engineering company retained by the user. Process objectives must be clearly communicated to ensure a successful outcome, but the potential for misunderstanding and miscommunication is high.
Because users want to “ensure that the catalyst will work under all conditions,” many request a design margin beyond what is necessary and then add another 20–30% to the normal feed rate. The catalyst vendor is committed to providing the requested capacity — 20% more flow requires 20% more catalyst, and the vendor will not complain about selling more catalyst. The specification of a more appropriate design margin presents an opportunity for optimization.
The concentrations of the key component in the feed and product have an impact on bed sizing, although not always in direct proportion (Step 6). The key component is either the contaminant that is to be removed or the reactant that is to be converted to some desirable product. The same precautions about generous design margins that apply to feed rate also apply to concentrations of key components. Higher feed concentration requires more catalyst. Higher concentrations may also affect the process configuration (Step 2) and the heat balance (Step 5).
The overall composition of the feed influences the calculated stream properties, such as density, heat capacity, viscosity, and surface tension. These properties, in turn, affect the calculated temperature rise (Step 5) and pressure drop (Step 7).
The most common error related to product specifications is a request for “zero” — e.g., no reactant in the reactor outlet or no byproducts. Zero concentration exists only in that it is below the current threshold of detection. The calculated contact time (and catalyst volume) is infinite if zero is used in most kinetic expressions and bed sizing methods (Step 6). A realistic and measurable target that is in line with final product specifications must be chosen. Tighter specifications, lower residual concentrations, and higher conversions increase catalyst bed size and thus cost.
The purity of the secondary reactant (hydrogen, oxygen, etc.) is reflected in the material balance and may have an effect on other aspects of the bed specification. Nonreactive material is a heat sink and reduces the calculated temperature increase (Step 5). The extra material can affect the secondary reactant concentration in the liquid phase, which may affect the reaction rate and the bed volume calculation (Step 6). The extra volume can also affect the fluid velocity and bed diameter calculation (Step 7). These effects are normally minor. The gas source pressure has a direct impact on the maximum operating pressure because adding a compressor is normally not an option.
The concentration of secondary contaminants (catalyst poisons) directly influences catalyst life and may affect the operating temperature and the bed sizing. The normal concentration should be based on multiple measurements over an extended time frame, not just the worst imagined concentration, because the calculated life (Step 8) is based on those values.
Each feedstock and operating condition can generate another case for specifying a catalyst bed. However, only the case requiring the largest bed is used to determine the final bed size. With few exceptions, the larger bed will work in the less-demanding cases, but a smaller bed will fail for the more-demanding case. The results of the bed sizing calculations for the less-demanding cases may be interesting and useful for optimization, but they should be considered later.
The engineering profession uses many different sets of units of measurement. (Even the SI system has common and formal variations.) The end-user, vendor, and engineering company must agree on a single set of units to reduce the potential for misunderstanding.
Step 2. Process configuration
The choice of reactor type is largely governed by the heat removal and hydraulic requirements of the process. Fortunately, most mature applications typically use only one or two types of reactors. Nevertheless, an understanding of the advantages and disadvantages of each type can help to validate the selection.
The most common reactors in commercial use are:
- batch slurry reactors
- continuous stirred-tank reactors (CSTRs)
- fixed-bed reactors, i.e., plug-flow reactors (PFRs)
- fluidized-bed reactors
- loop recycle reactors.
This article covers only the first three types of reactors because they are the most prevalent in the chemical process industries (CPI).
Batch slurry reactors are commonly used in labs and pilot plants in the discovery of new chemistry and catalysts. Finely divided (2–100 μm) powdered catalyst is combined with the bulk liquid in a stirred flask or tank to form a slurry. Gas introduced at the stirrer forms bubbles in the liquid. With care, the catalyst can be reused in many cases. Experimental results are easily reproduced and scaled up (within reason). Batch slurry reactors are favored by the pharmaceutical and fine chemicals industries.
The main drawbacks of batch slurry reactors are:
- There is a limit to the practical size of a batch reactor vessel, and there are no economies of scale when multiple vessels are required for the same duty.
- Batch time (and therefore reactor productivity) is the reaction time plus the time required to fill and discharge the reactor.
- The catalyst typically remains active (unless it is deactivated) during product discharge, creating additional reaction time that can change the product composition and byproduct formation.
- A product heel remains if the catalyst is reused, and product from one batch may contaminate the next batch.
Continuous stirred-tank reactors have many of the same operating characteristics as batch slurry reactors: the catalyst is a finely divided powder and gas forms bubbles in the liquid slurry. The key difference is the continuous addition of feed and withdrawal of product; catalyst may be periodically added and removed as well. The main advantages of a CSTR over a batch reactor are:
- no fill time
- no discharge time
- no product heel
- isothermal operation
- uniform product quality.
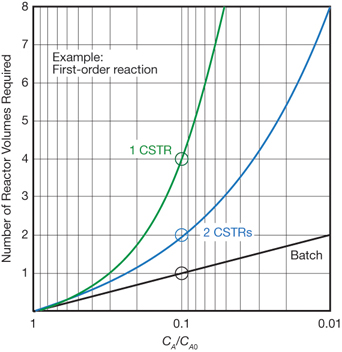
▲Figure 2. Achieving 90% conversion (CA/CA0 = 0.1) of a first-order reaction in two CSTRs (blue line) requires twice the reactor volume and twice as much catalyst as a batch reactor, while a single CSTR (green line) requires four times the reactor volume and four times the amount of...
Oligomerization rates are lower in a CSTR because reactant concentrations are generally lower in a CSTR than in a batch reactor. And if the reaction order is greater than zero, a larger reactor volume is needed. Continuous stirred-tank reactors require significantly more reactor volume — and catalyst — than a batch slurry reactor to achieve the same conversion (Figure 2).
Would you like to access the complete CEP Article?
No problem. You just have to complete the following steps.
You have completed 0 of 2 steps.
-
Log in
You must be logged in to view this content. Log in now.
-
AIChE Membership
You must be an AIChE member to view this article. Join now.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
